
In a sector where the right drug emerging at the right time can yield billions, Prescient Therapeutics Ltd (ASX:PTX, OTC:PSTTF) is working to become a success story, as its advanced T-Cell Lymphoma (TCL) drug, PTX-100, moves into Phase 2 clinical trials — where the most significant value inflection typically occurs in biotech.
According to Prescient, PTX-100 is showing promise in treating a brutal cancer with limited options, including a 64% reduction or halt in tumour growth in early-stage trials.
In a recent article, the company noted that the drug has already attracted key US Food and Drug Administration (FDA) designations — Orphan Drug and Fast Track — which could pave the way for PTX-100 to target a TCL market worth about US$1.8 billion, with a focus on Cutaneous T-Cell Lymphoma (CTCL).
The biotech formula: A strong product and a large market
In the biotech world, the ultimate questions are: can a product make it to market, and what kind of market awaits? As drugs progress through Phase 2, data readouts help determine whether a drug will succeed. This is where value inflection happens — when the market gains a clearer view of a drug’s potential.
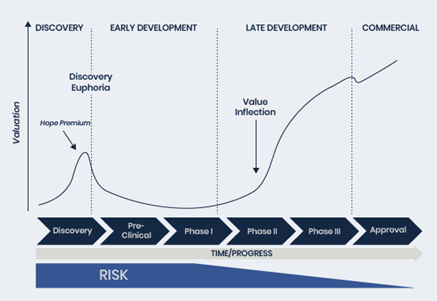
But getting approved is just part of the equation; the other key factor is the market size. The Orphan Drug Designation plays a pivotal role here, granting seven years of market exclusivity in the US (and 10 years in the EU) for drugs addressing rare diseases with unmet needs. According to Prescient, this recognition, combined with PTX-100’s clinical promise, positions the company to target a large, lucrative market for CTCL.
“PTX-100 is emerging as a standout candidate by ticking every box: a disease of unmet need, a large target market, promising trial results and recognition by the FDA, with the potential to apply to 22% of all cancer cases,” the company said.
Path to market: Exploring potential commercial opportunities
With more than 27,000 new TCL cases annually, PTX-100 is positioned to capture a significant share of the US$1.8 billion market. As the company progresses through Phase 2, three potential commercialisation paths are available, it said: acquisition, partnership or independent launch.
To illustrate how these paths could play out, Prescient looked at comparable case studies, such as Mirati Therapeutics and Dimerix. Mirati received Orphan Drug status and accelerated approval for its KRAS G12C mutation drug Adagrasib in 2022, leading to a $4.8 billion acquisition by Bristol Myers Squibb just 10 months later.
Similarly, Dimerix’s DMX-200, which is undergoing Phase 3 trials for the rare kidney disease focal segmental glomerulosclerosis (FSGS), secured multiple licensing deals worth millions, positioning it for future success.
“PTX-100 has already received Orphan Drug and Fast Track Designations from the FDA, which may expedite the development and approval processes, enhance its market visibility, and increase its attractiveness to potential acquirers,” Prescient said.
A blueprint for success: The future for PTX-100
Securing a Phase 2b registrational study for PTX-100 would put the drug in the ranks of high-value oncology assets, rendering it a highly attractive acquisition or partnership target. As Prescient noted, regulatory tailwinds combined with strong clinical data can command substantial value for late-stage oncology drugs.
With PTX-100’s outstanding Phase 1b results and FDA designations, Prescient is optimistic about its commercial opportunities, whether through partnerships, acquisition, or independent market launch.
link




